Ethyl benzoate, C9H10O2, is an ester formed by the condensation of benzoic acid and ethanol. It is a colorless liquid that is almost insoluble in water, but miscible with most organic solvents.
As with many volatile esters, ethyl benzoate has a pleasant odor described as sweet, wintergreen, fruity, medicinal, cherry and grape.[1] It is a component of some fragrances and artificial fruit flavors.
Preparation
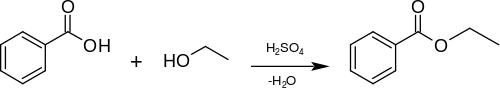
A simple and commonly used method for the preparation of ethyl benzoate in the laboratory is the acidic esterification of benzoic acid with ethanol and sulfuric acid as catalyst:[2]
References
- ^ Ethyl benzoate, thegoodscentscompany.com
- ^ Arthur Israel Vogel. Rev. by Brian S. Furniss: Vogel’s textbook of practical organic chemistry. 5. Auflage. Longman, Harlow 1989, ISBN 0-582-46236-3, S. 1076


Recent Comments