How Can We Help?
You are here:
< Back
The Algar–Flynn–Oyamada reaction is a chemical reaction whereby a chalcone undergoes an oxidative cyclization to form a flavonol.[1][2]
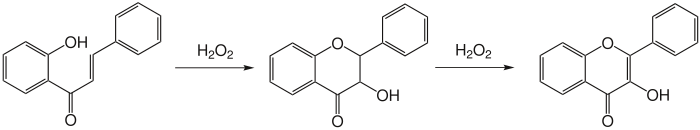
Reaction mechanism
There are several possible mechanisms to explain this reaction; however, these reaction mechanisms have not been elucidated. What is known is that a two-stage mechanism exists. First, dihydroflavonol is formed, which then subsequently oxidizes to form a flavonol.
Proposed mechanisms involving epoxidation of the alkene have been disproven.[3]
The probable mechanisms are thus two possibilities:
- The phenoxide attacks the enone at the beta position, and the alkene directly attacks hydrogen peroxide from the alpha position, forming the dihydroflavonol.
- The phenoxide attacks the enone at the beta position, closing the six-membered ring and forming an enolate intermediate. The enolate then attacks hydrogen peroxide, forming the dihydroflavonol.

See also
References
- ^ Algar, J.; Flynn, J. P. (1934). Proceedings of the Royal Irish Academy. 42B: 1.
{{cite journal}}: CS1 maint: untitled periodical (link) - ^ Oyamada, B. (1935). "A New General Method for the Synthesis of the Derivatives of Flavonol". Bulletin of the Chemical Society of Japan. 10 (5): 182–186. doi:10.1246/bcsj.10.182.
- ^ Gormley, T.R.; O'Sullivan, W.I. (1973). "Flavanoid epoxides—XIII". Tetrahedron. 29 (2): 369–373. doi:10.1016/S0040-4020(01)93304-6. hdl:10197/6996. ISSN 0040-4020.
Categories
-
Annuals36
-
Bulbs, Corms & Tubers41
-
Ferns27
-
Fruits3
-
Garden Plants23
-
Grasses26
-
Herb17
-
Insects1
-
Mammals1
-
Midwest Native Plants0
-
Northeast Native Plants112
-
Perennials123
-
Rose1
-
Shrubs47
-
Trees112
-
Tropical Plants53
-
Upland Birds5
-
Vines18
-
Viola Tricolor1
-
Water Gardening & Plants9
-
Waterfowl0
-
Wetland Birds0
-
Wetland Plants4
-
Wildbirds172
-
Wildflowers1
-
Woodland Plants29
Table of Contents


Recent Comments