Vanadium(IV) fluoride (VF4) is an inorganic compound of vanadium and fluorine. It is paramagnetic yellow-brown solid that is very hygroscopic.[2] Unlike the corresponding vanadium tetrachloride, the tetrafluoride is not volatile because it adopts a polymeric structure.[5] It decomposes before melting.
Preparation and reactions
VF4 can be prepared by treating VCl4 with HF:
- VCl4 + 4 HF → VF4 + 4 HCl
It was first prepared in this way.[6]
It decomposes at 325 °C, undergoing disproportionation to the tri- and pentafluorides:[2]
- 2 VF4 → VF3 + VF5
Structure
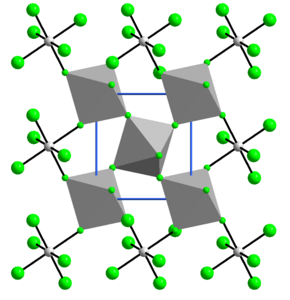
The structure of VF4 is related to that of SnF4. Each vanadium centre is octahedral, surrounded by six fluoride ligands. Four of the fluoride centers bridge to adjacent vanadium centres.[7]
References
- ^ a b c d e Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ^ a b c d Kwasnik, W. (1963). Brauer, Georg (ed.). Handbook of Preparative Inorganic Chemistry (UK ed.). London: Academic Press. pp. 252–253.
- ^ a b c Anatolievich, Kiper Ruslan. "vanadium(IV) fluoride". chemister.ru. Retrieved 2014-06-25.
- ^ "Vanadium(IV) fluoride, 95%". alfa.com. Alfa Aesar. Retrieved 2014-06-25.
- ^ Cotton, F. Albert; Wilkinson, Geoffrey; Murillo, Carlos A.; Bochmann, Manfred (1999), Advanced Inorganic Chemistry (6th ed.), New York: Wiley-Interscience, p. 716, ISBN 0-471-19957-5
- ^ Otto Ruff, Herbert Lickfett "Vanadinfluoride" Chemische Berichte 1911, vol. 44, pages 2539–2549. doi:10.1002/cber.19110440379
- ^ Becker S., Muller B. G. Vanadium Tetrafluoride, Angew. Chem. Intnl. Ed. Engl. 1990, vol. 29, page 406


Recent Comments